Glenmark plans to launch Fabiflu 400 mg for the treatment of Covid-19:
Pharma News – 9 August: Glenmark Pharmaceuticals announced the launch of Fabiflu 400 mg in the Indian market (On 6 August 2020). Fabiflu is an oral antiviral drug that was initially launched in the 200 mg strength. It is used for the treatment of mild to moderate Covid-19 patients.
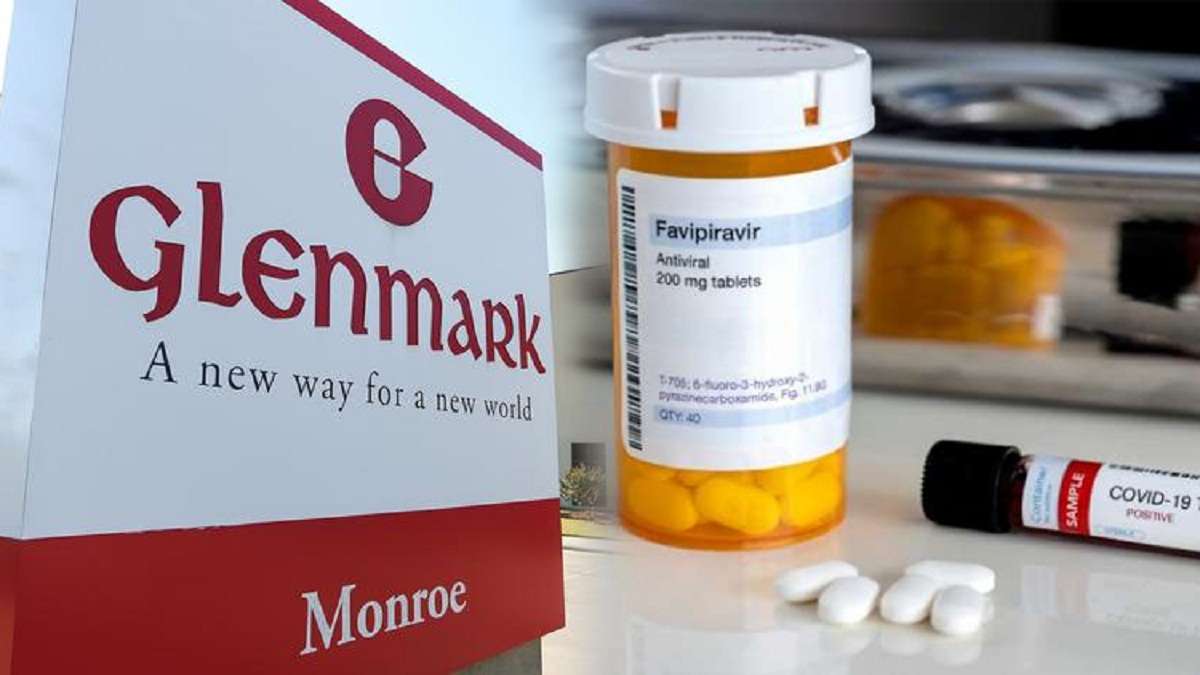
The drug firm claimed that the higher dose would be beneficial in treating Covid-19 patients. It is also believed to improve patient compliance by reducing the number of tablets consumed daily. The company also added that only 9 tablets of Fabiflu 400 mg need to be taken on Day 1. This is split up as 4.5 tablets each in the morning and evening of the first day. From Day 2, each patient must consume 2 tablets twice a day until the end of the course.

Glenmark Pharma is the first company to receive regulatory approval for the sale of 400 mg dosage form in India.
It must be noted that the 200 mg dosage was developed in accordance with the global formulation of Favipiravir 200 mg. The 400 mg version, however, was developed by Glenmark’s R&D[1] entirely and did not depend on any existing global formulation.
The post-marketing surveillance of Fabiflu is currently being done on a large pool of patients. This is done to establish the safety and efficacy of the oral antiviral Favipiravir.
Abbreviation : 1. Research and Development
Lupin signs an exclusive pact with ForDoz for the distribution of 2 complex injectable drugs in the US:
Pharma News – 9 August: The US[1]-based subsidiary of Lupin Ltd has signed an exclusive pact with ForDoz Pharma Corp on 6 August 2020. This was signed to facilitate the distribution of two complex injectable drugs in the US.

One of the drug candidates belongs to the oncology segment while the other to the anti-infective segment. Both are expected to be affordable alternatives to the existing drugs.
As per the pact, ForDoz will be responsible for the development, production and receiving of regulatory approval of the products. The company’s manufacturing unit in the US will be involved in all these processes. Lupin, on the other hand, is expected to take care of the marketing and distribution of these drug candidates throughout the country.
Lupin also believes that this partnership will aid in its organic R&D[2] of long term injectables and other biosimilars. ForDoz too will benefit from this partnership as it now has access to Lupin’s marketing and distributing network.
The financial details of this pact are yet to be disclosed as both the pharmaceutical companies have remained tight-lipped on this matter.
Lupin has expanded its portfolio from simple generic products to speciality products by making use of the increasing competition. Production of speciality generics and biosimilars have increased the profit margin of the company.
To date, Lupin has launched only a handful of specialty generics. This includes women’s health drugs like Methergine, Solosec and cholesterol-lowering drugs like Antara. The drug firm is also working continuously for the launch of other specialty drugs like ProAir and Brovana. ProAir is a drug used for asthma. Brovana is an inhalation solution that is also used in the treatment of asthma.
Abbreviation :
- 1. United States
- 2. Research and Development
3. Disulfiram has the potential to combat against SARS-CoV-2 by acting on the Mpro:
Pharma News – 9 August: The Researchers from National Research University HSE[1], Russia have identified a potential drug to combat SARS-CoV-2[2]. According to these scientists, the drugs used to treat alcoholism could have a significant impact on Covid-19 patients.

They revealed that the structural elements of SARS-CoV-2 that are less susceptible to mutations are better targets for potential drug candidates. This includes conservative proteins like Mpro [3] that plays an important role in virus replication. Inhibition of this protein might either slow down or completely stop the reproduction of the coronavirus in the host body.
Modeling studies showed that the active center of Mpro has high ligand efficiency with sulfur-containing drugs. Among the various drugs obtained from the USFDA[4], disulfiram was found to the most effective.
Disulfiram is used for treating alcoholism but this drug has stable interactions with the Mpro of SARS-CoV-2. This drug was found to act in two ways. It acts as a covalent inhibitor of the virus and counteracts the decreased levels of reduced glutathione in the body.
.jpg)
The Russian scientists also claimed that neratinib, an irreversible tyrosine kinase inhibitor, has some potential action against SARS-CoV-2.
The tests to confirm the efficacy of disulfiram was conducted on 27 July 2020. The results of this test revealed that disulfiram inhibits Mpro in 100 nm concentration. This however did not validate the hypothesis regarding neratinib as it was found to be ineffective in clinical use.
Abbreviation :
- 1. Higher School of Economics
- 2. Severe Acute Respiratory Syndrome- Coronavirus – 2
- 3. Main protease
- 4. United States Food and Drug Administration
4. India could strategically benefit from Trump’s “Buy American first” pharma executive order :
Pharma News – 9 August: On 6 August 2020, Donald Trump stated that the United States (US) must become self-reliant in terms of pharmaceuticals. He further added that this was the need of the hour as the US was highly dependent on nations like China for the supply of essential drugs.
His statements were supported by the “Buy American first” pharma executive order. This order was passed to ensure the development of API[1] and other critical medicine in the US.
While this may be a boon to the US pharma firms, its effect on the Indian pharma companies is quite diversified.
Many claim that this move might be a strategic positive for India as this would completely stop the import of API to the US from China. Due to cost differential in the manufacturing of generic drugs, many believe that it would be difficult to move sizable manufacturing to the US. This might implore that India could take part in the manufacturing of generics as it holds a major share in the manufacturing of generics globally.
Experts claim that India and the US have a complementary relationship in terms of the pharma sector. Major pharmaceutical industries like Cipla, Sun Pharma and Dr.Reddy’s among others are present in the US.
This order will drastically affect the API manufacturers from China as China is the world’s largest source of API. It must be noted that India also imports a major portion of its API from China.
Abbreviation : 1. Active Pharmaceutical Ingredient
Read more:
- Pharma news – 20 June,2020
- Pharma News Updates – 23 July 2020
- Pharma News 16 July 2020 Updates
- Pharma News – 13 August 2020
References:
- [1] – Business Standard – 6 August 2020
- [2] – Live mint – 6 August 2020
- [3] – Business Today – 7 August 2020
- [4] – DNA India – 7 August 2020


Pingback: Pharma Trends and News Update: 23 Aug' 2020 > PharmaCampus
Pingback: Pharma news – June 20 Updates > PharmaCampus