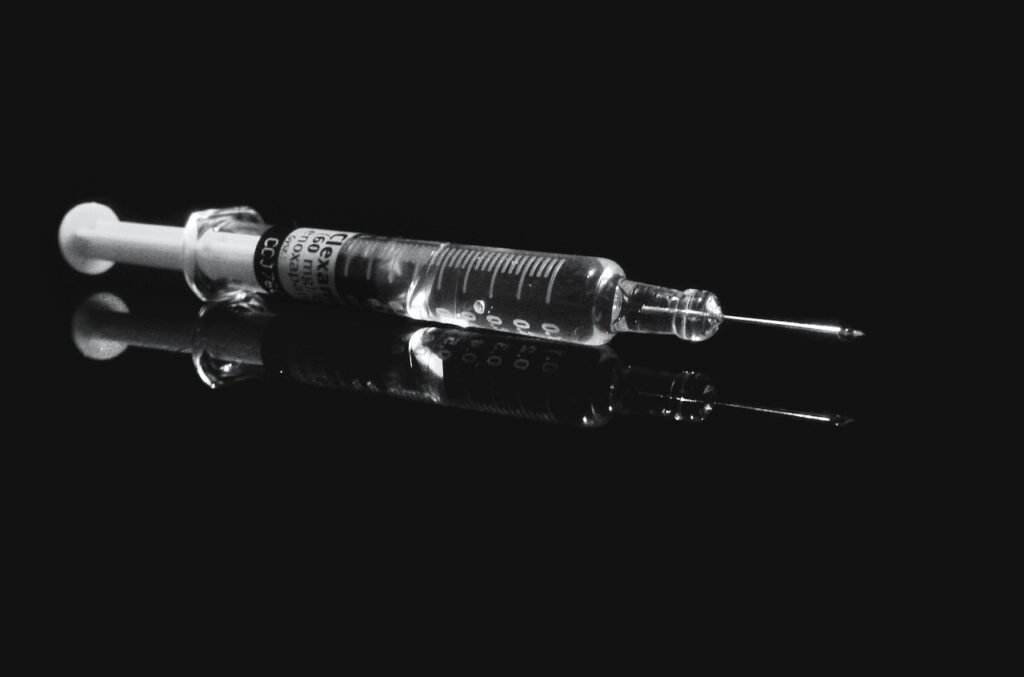
USFDA approves Gland Pharma’s generic Foscarnet Sodium Injection, single – dose bag for infusion :
Gland Pharma had sought out the USFDA’s[1] approval for one of its generic products. This product is called Foscarnet Sodium injection, 6000 mg/ 250 mL ( 24 mg/ mL ) Single-Dose Bag for Infusion.
On 22 April 2021, the company announced that the FDA had approved this product. This approval has led to Gland Pharma securing the first generic offering status in Single – Dose Bag for Infusion. What is the role of USFDA in protecting health?
This generic product is bioequivalent and therapeutically equivalent to Foscarnet Injection, 24 mg/ mL. Clinigen Healthcare manufactures this Reference Listed Drug (RLD). [3]
About Foscarnet Sodium Injection
The USFDA has granted CGT[3] designation for Foscarnet Sodium Injection. This is because it is the first approved applicant for such competitive gene therapy. Per the FDA norms, Gland Pharma is eligible for 180 days of CGT exclusivity that will begin after the commercial launch date of the product.
Gland Pharma has an in–house API [2] Source for this product and plans to supply it globally. The global market size of this product is estimated to be around $ 50 million. The drug firm has also launched this product in the Canadian market. Read also: Gland Pharma agreement with RDIF for Sputnik V.
This injection is used to treat cytomegalovirus ( CMV ) retinitis in patients with AIDS[4]. It is also indicated for treating acyclovir–resistant mucocutaneous herpes simplex virus ( HSV ) infections. This drug is especially used in treating such infections in immunocompromised individuals.
Per IQVIA[5], the US sales of this injection and its generic equivalents for 12 months ending on January 2021 is around USD[6] 28 million. Know about IQVIA- The Human Data Science Company.
Gland Pharma is one of the largest injectable–focused companies specializing in small volume liquid parenteral products. It manufactures a wide range of products, including vials, ampoules, infusions, oncology, and ophthalmic solutions. It has also pioneered Heparin technology in India.
Abbreviation :
- 1. United States Food and Drug Administration
- 2. Active Pharmaceutical Ingredient
- 3. Competitive Gene Therapy
- 4. Acquired Immunodeficiency Syndrome
- 5. Formerly known as Quintiles and IMS Health Inc.
- 6. United States Dollar
Explore other Articles
- Pharma News – 23.01.2023 to 29.01.2023
- Lupin issues nationwide recall of Clobetasol propionate cream.
- Global Pharma Healthcare recalls Artificial Tears Lubricant eye drops.
- Milestones in Cancer Research and Discovery
- OAI notice for Torrent Pharma Indrad facility
- Cancer Cachexia: Mankind Pharma’s association with Actimed Therapeutics